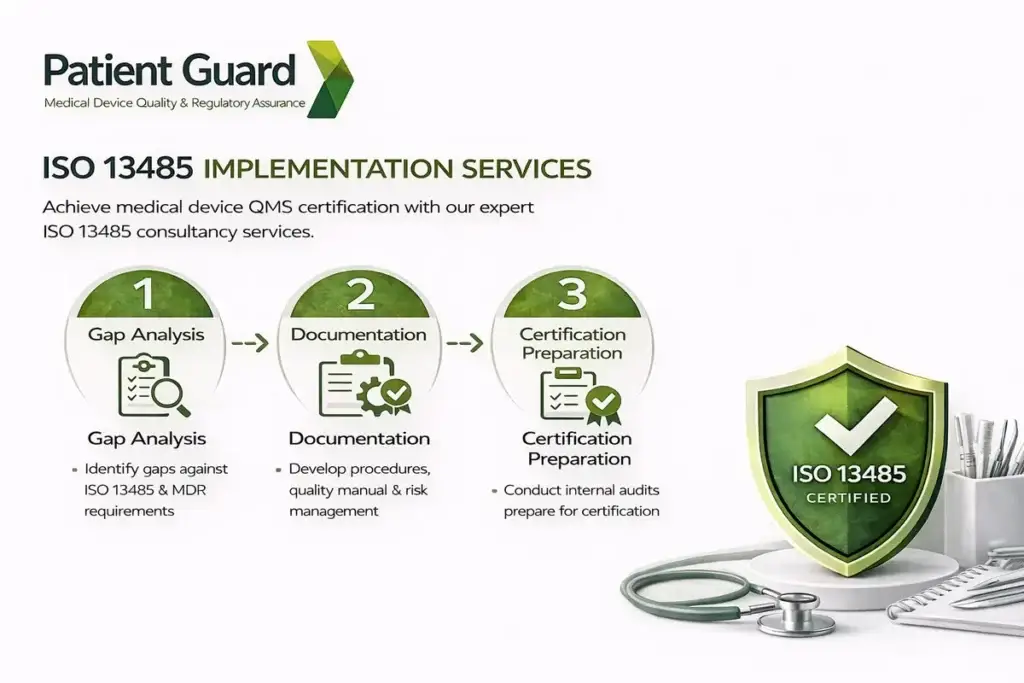
ISO 14971 Risk Management Implementation Guide
Medical devices exist to improve health outcomes, but every device carries potential risk. Managing those risks in a structured, documented, and defensible way is essential for regulatory approval and patient safety.