
The MHRA 2026 Regulatory Roadmap Explained
Learn how the MHRA 2026 Regulations will impact UKCA compliance, IVD classifications, AI medical devices, IRP pathways, and post-market surveillance.
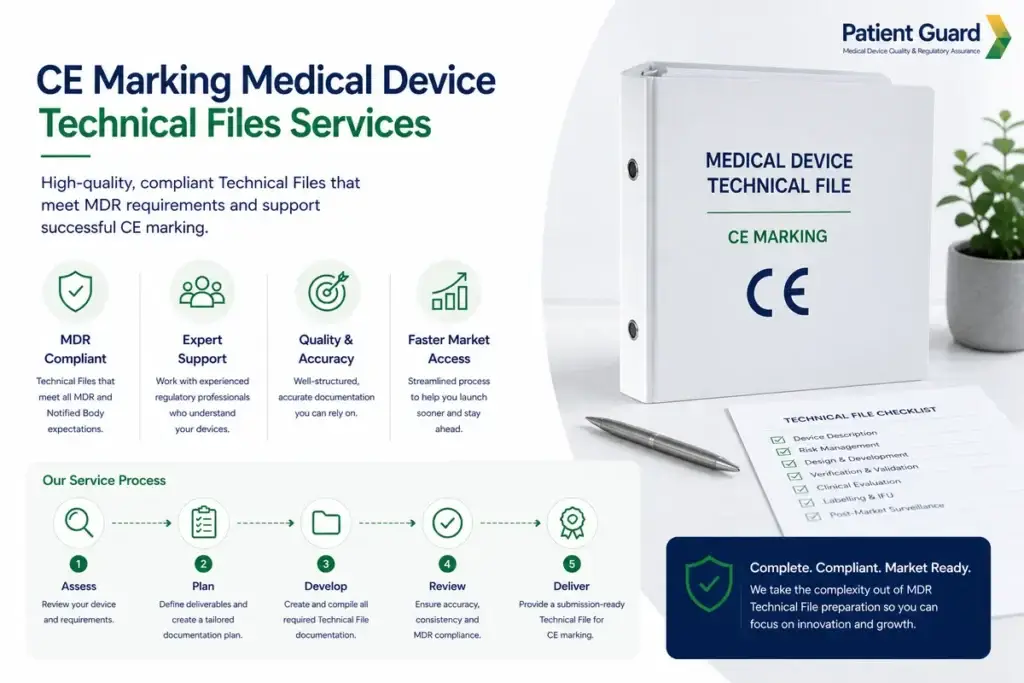
Expert compilation, review, and remediation of EU MDR 2017/745 Technical Documentation. Ensure your dossier is audit-ready for Notified Body submission

Medical device manufacturers placing products on the European market must prepare and maintain comprehensive technical documentation to demonstrate compliance with the EU Medical Device Regulation (MDR) 2017/745.
A CE Marking Technical File is a core regulatory requirement for medical devices and provides the documented evidence needed to demonstrate that a device meets the applicable General Safety and Performance Requirements (GSPRs).
Patient Guard provides expert CE Marking Technical File services for medical device manufacturers, supporting the preparation, review, remediation, and ongoing maintenance of MDR-compliant technical documentation.
We help manufacturers build structured, audit-ready Technical Files that align with MDR Annex II and Annex III requirements and support notified body review where applicable.
Whether you are preparing a new Technical File, updating legacy documentation, or addressing identified gaps, we ensure a clear and compliant route to CE marking readiness.


Our consultants are medical device specialists who confidently guide you through the strict requirements of EU Regulation 2017/745.
From Class I self-certified devices to complex Class III implantables, we have extensive hands-on experience authoring technical files for all risk tiers.
We build and format technical documentation to align perfectly with the precise expectations, terminology, and assessment pathways of top Notified Bodies.
We don’t believe in over-complicating compliance. We build clear, lean technical documentation frameworks customized for your specific product line.
We hook your technical files cleanly into your active EN ISO 13485 Quality Management System, risk data, and clinical evidence streams.
We structure your documentation with proactive Post-Market Surveillance (PMS) and PMCF systems built-in, making long-term maintenance easy.
Drafting precise device descriptions, intended use justifications, basic UDI-DI setup, and formal MDR classification rationales.
Compiling the mandatory checklist demonstrating compliance with all relevant General Safety and Performance Requirements.
Authoring or auditing your risk files, hazard lists, mitigation steps, and final benefit-risk analyses to establish product safety.
Organizing and presenting physical testing metrics, electrical safety (IEC 60601), biocompatibility data, and sterilization validations.
Developing robust Clinical Evaluation Plans and Reports (CEP/CER) that verify your device performance using solid clinical data.
Reviewing packaging layouts, caution symbols, medical device identification markers, and technical instructions for use (IFU).
Under Regulation (EU) 2017/745, manufacturers must prepare and maintain technical documentation that demonstrates conformity with the applicable requirements of the MDR.
A compliant Technical File for a medical device typically includes:
| No | Heading | Contents |
|---|---|---|
| 1. | Device Description | Full specifications, intended use, and UDI-DI |
| 2. | Labelling & IFU | Instructions for use in all required EU languages. |
| 3. | Design & Manufacturing | Documentation of design stages and supply chain sites. |
| 4. | GSPR Mapping | Evidence for General Safety and Performance Requirements. |
| 5. | Benefit-Risk Analysis | Full risk management files (ISO 14971) |
| 6. | Verification & Validation | Bench testing, sterilization, biological evaluation, clinical evaluation |
| 7. | Post Market Surveillance | PMS Plan, PSUR, PMCF Plan, PMCF Reports |
Is your CER MDR-compliant? Under the latest MDR standards, clinical evidence must be proactive and continuous. We ensure your Technical File contains a robust Clinical Evaluation Report supported by your Post-Market Surveillance (PMS) data.
If your device incorporates AI or machine learning, your technical documentation must now address the requirements of the EU AI Act. We specialize in mapping software lifecycle processes (IEC 62304) and cybersecurity measures into your CE dossier.
We assess your device, classification, and current documentation status.
We review your existing Technical File or source documents against MDR requirements.
We identify missing sections, weak justifications, and documentation gaps.
We help compile, revise, and structure the Technical File for compliance.
We support final checks ahead of submission, audit, or notified body review.
We support Technical File preparation for a wide range of medical devices, including:
From
Ensure regulatory compliance and market readiness with expertly prepared CE Marking Medical Device Technical Files. Pricing starts from £6,000, with preferential rates for multiple products.
Assessment of your device and existing documentation against MDR Annex II & III requirements.
Compilation, structuring, and correction of documentation including GSPR, risk management, and clinical data.
Final compliance check and preparation for Notified Body review or EU market placement.
Fast turnaround: most Technical Files are delivered within 4–8 weeks, subject to completeness of product information.
A CE Marking Technical File is the set of technical documentation required to demonstrate that a medical device complies with the applicable requirements of the EU Medical Device Regulation (EU) 2017/745.
Yes, all medical devices placed on the EU market require technical documentation. The scope and complexity depend on the device classification, intended purpose, and applicable conformity assessment route.
A Technical File typically includes device description, design and manufacturing information, risk management, GSPR compliance, verification and validation data, clinical evaluation, labelling, and post-market surveillance documentation.
Yes, we can review your current Technical File, identify compliance gaps, and help remediate the documentation to align with MDR requirements.
Timelines depend on the complexity of the device and the availability of supporting documentation. Where the source documentation is already available, review and remediation can begin immediately.
Click on the links below to discover more:

Learn how the MHRA 2026 Regulations will impact UKCA compliance, IVD classifications, AI medical devices, IRP pathways, and post-market surveillance.

Explore medical device regulatory consulting services for ISO, FDA, EU, and more. Build compliant systems, pass audits, and scale globally with expert support.

A milestone that strengthens our commitment to quality
Regulatory and quality consulting is one thing. Building, implementing, and passing audits on your own Quality Management System is another.

UDI medical devices are not just a labelling requirement. They are the backbone of traceability under EU MDR.

EUDAMED enters a new phase as four modules become mandatory from 28 May 2026. Learn what changes and how to prepare for compliance.

Medical devices exist to improve health outcomes, but every device carries potential risk. Managing those risks in a structured, documented, and defensible way is essential for regulatory approval and patient safety.
Speak to one of our regulatory and compliance experts to arrange an obligation-free call. Our experienced team is ready to help you get your medical device to market.
UK Office
For help with the checklist or other aspects of your compliance journey, please reach out to us at Patient Guard and our experts would be happy to help.
UK Office
Thank you! The checklist is now ready to download.

For help with the checklist or other aspects of your compliance journey, please reach out to us at Patient Guard and our experts would be happy to help.
UK Office