Introduction
It’s a common assumption: if you’re ISO 9001 certified, you’re already compliant with ISO 13485. Unfortunately, that’s one of the most expensive misunderstandings a medical-device company can make.

While both standards share the same DNA, they serve very different purposes. ISO 9001 is the universal language of quality management; ISO 13485 is its medical-device-specific dialect – one that speaks directly to regulators, auditors, and patients.
The stakes are high. Choosing the wrong framework or failing to transition properly can delay certification, stall market access, and attract unwelcome audit findings.
Get expert support upgrading from ISO 9001 to ISO 13485 with Patient Guard – guiding you from business quality to regulatory excellence.
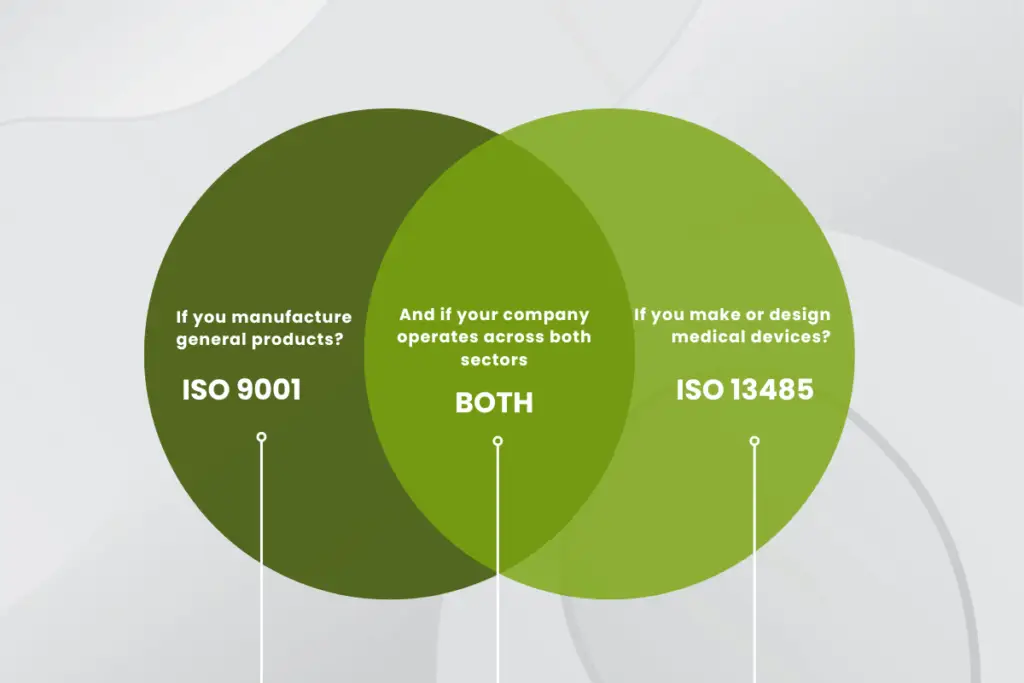
ISO 9001 vs ISO 13485 – The Overview
What Each Standard Covers
Feature | ISO 9001:2015 | ISO 13485:2016 |
|---|---|---|
Industry Scope | Universal – applies to all sectors | Medical devices and related services |
Primary Objective | Customer satisfaction and process improvement | Patient safety and regulatory compliance |
Improvement Focus | Continual improvement (PDCA cycle) | Maintaining stable, validated processes |
Risk Approach | Business and operational risk | Product safety and clinical risk (aligned with ISO 14971) |
Regulatory Alignment | None required | Supports MDR, IVDR, and FDA QMSR |
Documentation Flexibility | Lean and adaptable | Mandatory traceability and record control |
Training & Competence | Encouraged | Required and verified |
Intended Outcome | Efficient, profitable operations | Safe, compliant medical devices |
Both standards follow the same Plan-Do-Check-Act (PDCA) logic and require documented processes, leadership commitment, and continual improvement.
However, ISO 13485 adds the clinical dimension that ISO 9001 deliberately omits. It turns quality from a business discipline into a safety system.
According to the ISO Survey 2023, there were approximately 837,052 valid ISO 9001 certificates worldwide, compared with just 32,963 ISO 13485 certificates, underscoring how much narrower and more specialised the medical-device standard remains. Europe alone accounts for roughly 40% of all management-system certifications, making the EU and the UK key hubs for ISO 13485 adoption (CertiGet EU, 2024).

The Main Differences Between ISO 9001 and 13485
Purpose and industry focus
ISO 9001 drives customer satisfaction and process optimisation across any industry. ISO 13485, by contrast, embeds medical-device-specific safety and regulatory controls. One measures success by efficiency; the other by patient outcomes.
Image #1: Quality manager comparing two QMS frameworks (pain/problem).
Risk and regulatory requirements
ISO 9001 treats risk as a business consideration, including supply disruptions, delivery times, and profitability. ISO 13485 goes deeper, integrating risk management under ISO 14971, which covers product hazards, clinical evaluation, and post-market vigilance.
This regulatory linkage is what makes ISO 13485 essential for MDR and FDA compliance.
Documentation and record control
ISO 9001 allows flexibility in documentation. ISO 13485 demands meticulous traceability: every device, component, and test record must be documented, version-controlled, and retrievable. “Good enough” document control under ISO 9001 rarely survives an MDR audit.
Continuous improvement vs compliance stability
ISO 9001 emphasises continual improvement, expecting organisations to evolve constantly. ISO 13485 values stable compliance, like maintaining validated processes that consistently produce safe, conforming devices. Improvement happens, but never at the expense of validation integrity.
Integration with other regulations
ISO 13485 serves as the quality backbone for the EU MDR, IVDR, and the FDA’s QMSR Final Rule (2024). For medical-device companies, it’s the regulatory passport.
You may also be interested in our article, Mastering ISO 13485 Compliance With a Lean QMS.
When to Choose ISO 9001 or ISO 13485
For general manufacturers, ISO 9001 remains the ideal standard, as it drives operational consistency, cost efficiency, and customer satisfaction.
For medical device and in vitro diagnostic manufacturers, however, ISO 13485 is non-negotiable. It demonstrates regulatory compliance, product traceability, and patient-safety controls.
Suppliers in the device value chain often hold dual certification, combining ISO 9001 for broad business clients and ISO 13485 for regulated markets.
According to Cognitive Market Research (2024), Europe accounts for more than 30% of the global ISO-certification market, and the UK alone generated approximately USD 517 million in 2024, growing at a projected 14.3% CAGR. This sustained growth signals that organisations view quality certification not as a cost, but as an investment.
Transitioning from ISO 9001 to ISO 13485
If you already have ISO 9001, you’re halfway there. Transitioning involves adding regulatory rigour and clinical risk management to an existing QMS.
Step-by-step path:
- Gap assessment – Identify where ISO 9001 processes fall short of 13485 clauses.
- QMS upgrade – Add required procedures: vigilance, device-master records, and traceability.
- Risk integration – Align with ISO 14971 methodology.
- Internal audit – Validate changes through impartial review.
- Certification audit – Engage a Notified Body or accredited registrar.
Transition projects typically run three to six months, depending on company size, but the benefits of market access, regulatory confidence, and fewer audit findings are immediate. Speak to one of our experts about our ISO 13485 transition and compliance services. Get in touch.
Combining ISO 9001 and ISO 13485 for a Lean QMS
A Lean QMS approach can effectively merge the two frameworks. Both share foundational processes, such as management review, internal audits, supplier control, and training, so duplication is unnecessary.
By integrating overlapping procedures and maintaining a unified document structure, companies gain clarity and reduce maintenance effort.
This hybrid model works exceptionally well for component suppliers and contract manufacturers serving both regulated and non-regulated sectors.
The result: less paperwork, more compliance.
Upgrade from ISO 9001 to ISO 13485 with Patient Guard – streamlined compliance, stronger credibility, zero confusion. Speak to one of our team members today. Get in touch.
Common Misconceptions About ISO 9001 vs ISO 13485
Common Misconceptions About ISO 9001 vs ISO 13485
- “They’re the same.”
False. ISO 13485 builds on ISO 9001, adding regulatory, traceability, and validation requirements. - “ISO 9001 covers medical devices.”
False. Regulators require ISO 13485 or equivalent. - “ISO 13485 doesn’t need improvement.”
False. Continual improvement still applies within validated boundaries.
Key Takeaways for Quality Managers
ISO 9001 and ISO 13485 are complementary, not competing, frameworks. The key is aligning them with your business model: efficiency versus compliance, customer versus regulator.
A quick summary for decision-makers:
Aspect | ISO 9001 | ISO 13485 |
Purpose | Process and customer satisfaction | Regulatory compliance and patient safety |
Applicability | All industries | Medical devices and IVDs |
Improvement Focus | Continual improvement | Maintaining validated processes |
Risk Model | Operational | Product and clinical |
Documentation | Flexible | Strict traceability |
Audit Frequency | Typically annual | Driven by the regulatory cycle |
Integration with MDR/FDA | None | Full alignment |
Understanding these contrasts helps leadership teams allocate resources wisely—building lean systems that satisfy both regulators and business partners.
Conclusion
In the debate of ISO 9001 vs ISO 13485, the winner depends on your market. ISO 9001 strengthens operations; ISO 13485 safeguards patients and market approval.
For medical-device companies, ISO 13485 isn’t just preferable, it’s essential for MDR audit readiness and global recognition. For others in the supply chain, combining both can unlock broader contracts and enduring trust.
Contact Patient Guard to transition smoothly from ISO 9001 to ISO 13485 and develop a Lean QMS that’s ready for every regulator. Talk to our experts.
Frequently Asked Questions
ISO 9001 is a generic quality management standard; ISO 13485 is specific to medical devices and focuses on patient safety, regulatory requirements, and traceability.
Most require ISO 13485 to meet MDR and FDA expectations. Some maintain ISO 9001 certification as well if they supply products outside the medical device domain.
With an existing ISO 9001 foundation, most companies can transition within a few months by adding risk management, validation, and documentation controls.
ISO 13485 aligns directly with MDR requirements, covering design control, post-market surveillance, and vigilance—making it the standard auditors look for first.
Yes. A unified Lean QMS can serve both, reducing duplication and audit effort while maintaining distinct compliance clauses.
Absolutely. Patient Guard provides gap assessments, transition planning, and internal-audit support to help your organisation achieve seamless compliance.
Patient Guards Recent Posts

ISO 14971 Risk Management Implementation Guide
Medical devices exist to improve health outcomes, but every device carries potential risk. Managing those risks in a structured, documented, and defensible way is essential for regulatory approval and patient safety.

Clinical Evaluation Report: EU MDR Requirements
Clinical evidence is central to demonstrating the safety and performance of medical devices in the European Union.

Medical Device Labelling Requirements Explained
Medical device labelling is more than a packaging exercise. It is a regulatory requirement that communicates essential information about a device’s identity, safety, and intended use.




