Medical Device Design and Development
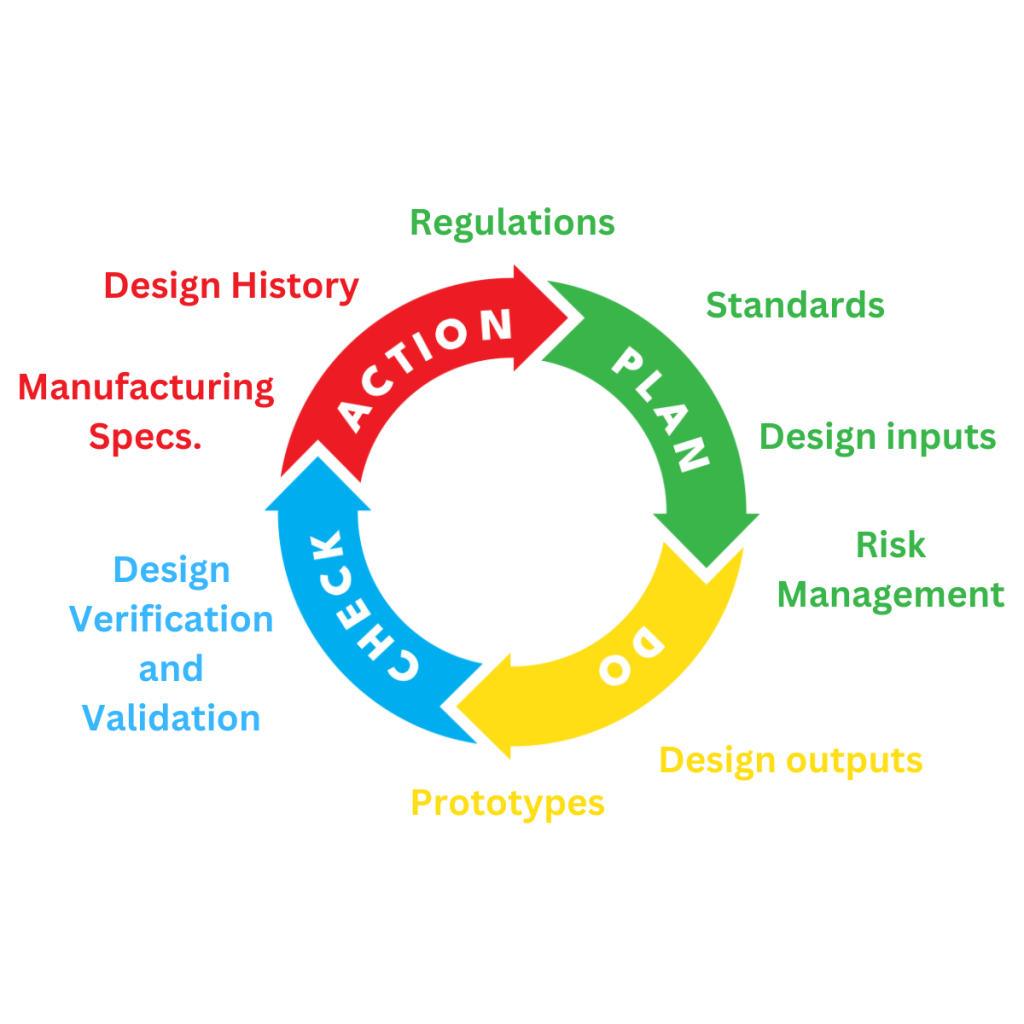
Planning for the design and development of a medical device is a requirement of regulatory systems. All manufacturers of Medical Devices are required to maintain a Quality Management System, in the USA this is determined under the CFR Title 21 part 820. Within the European Union, it is placed within demonstrating certificated compliance with international standard EN ISO 13485. The USA has recently indicated that they will also use the ISO 13485 standard for the requirements of manufacturers being compliant for QMS purposes.
Medical Device Design and Development Read More »